Over a quarter of a century before the FDA’s “Patient-Focused Drug Development” became a critical goal in drug and device approvals, Phase V had trademarked the term “Phase V®” to describe patient-centered outcomes as the “fifth phase of therapeutic testing” in the drug and device industries.

Patient-Centered Outcomes - Since 1987
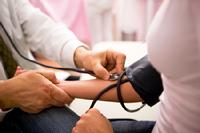
direct-from-patient health outcomes
Providing Digital Health Technologies for Remote Acquisition in Clinical Investigations

Analytics - how we get it right
a boutique clinical research service provider

Patient-Focused Drug Development
With Phase V’s focus beyond laboratory and clinical assessment, Phase V emphasizes the importance of demonstrating the effectiveness of therapies and medical interventions in real world settings from the patient’s perspective.
To achieve the real-world delivery of patient data, since 2004, Phase V has used device enabled technologies to incorporate direct-from-patient data to the clinical research studies for new drugs and devices during pre-marketing and post-marketing studies.
Our Services
40 Years of Scientific Innovation in Direct-from-Patient Assessments
Phase V Technologies was built on a strong foundation of scientific innovation in clinical trial research, promoting new methods for therapeutic drug and device development.40 Years of Regulatory of Patient Reported Outcomes Assessment
Phase V Technologies was the first Contract Research Organization to promote the use of patient-reported outcomes including the assessment of quality of life and treatment satisfaction in pivotal Phase 3 clinical tirals.40 Years of Advancing Technological Development
Phase V Technologies has excelled in advancing technology in the measurement, data collection, intensively longitudinal big data analytics and statistical analyses as part of its Phase V® Outcomes Information System for clinical trials research.A sampling of diabetes studies over the years
799-P: Gender Differences in Diabetes Distress Comparing Dapagliflozin plus Saxagliptin to Glimepiride When Added to Metformin in Poorly Controlled Type 2 Diabetes
MARCIA A. TESTA and DONALD C. SIMONSON, Boston, MA Gender Differences in Diabetes Distress Comparing Dapagliflozin plus Saxagliptin to Glimepiride When Added to.
810-P: Patient-reported outcomes in T2D patients inadequately controlled by metformin are more favorable for dapagliflozin plus saxagliptin vs. insulin glargine.
MARCIA A. TESTA, DONALD C. SIMONSON, ELLA EKHOLM, MAXWELL SU, EVA K. JOHNSSON, Boston, MA, Mölndal, Sweden Patient-Reported.
1520-P: Impact of Patient-Centered Factors on Comparative Effectiveness of Initial Therapies in Treatment-Naïve T2D
MARCIA A. TESTA, ALEXANDER TURCHIN, MAXWELL SU, DONALD C. SIMONSON, Boston, MA Impact of Patient-Centered Factors on Comparative.